News from Cambridge University shows some promise for inexpensive production of hydrogen, an elusive process considering the lightest element in creation is also the most common, said to make up 90 percent of the visible universe. On earth, it readily combines with oxygen to form water, a handy thing to have around for the benefit of our species.
Getting hydrogen out of the water so that we can burn it in our cars and airplanes is a frustrating process, though, often requiring more energy for the extraction than can be obtained from its combustion.
According the National Renewable Energy Laboratory, “To make [hydrogen] usable in fuel cells or otherwise provide energy, we must expend energy or modify another energy source to extract it from the fossil fuel, biomass, water, or other compound in which it is found. Nearly all hydrogen production in the United States today is by steam reformation of natural gas. This, however, releases carbon dioxide in the process and trades one relatively clean fuel for another, with associated energy loss, so it does little to meet national energy needs.”
The NREL discusses four other methods of producing hydrogen, but each ends up costing too much for the energy created, or emits CO2 or other gases that may be more harmful than the resulting hydrogen isbeneficial in the long term.
Thermochemical hydrogen comes from heating biomass or fossil fuels with little or no oxygen present and creating synthetic gas or “syngas.” This syngas can be catalytically converted to increase the amount of hydrogen, but still with gaseous emissions.
Electrolysis can split water into hydrogen and oxygen, reversing the reaction in a fuel cell. The NREL points out the process must use an inexpensive source of electricity such as wind to make economic sense. Wind’s variability limits its use for full-time hydrogen production, however.
Electrochemical photolytic (PEC) hydrogen production combines electrolysis with photovoltaic (PV) electricity to provide the necessary energy for splitting water. This direct process is efficient, but requires PV materials “strong enough to drive the water split and stable in a liquid system,” two essentials apparently difficult to achieve with current materials.
Biological Photolytic Hydrogen emulates nature, using photosynthesis to make hydrogen instead of sugar and oxygen. On the surface, this seems exceedingly green, but the catch here is that the algae and photosynthetic bacteria used to produce hydrogen don’t work well in an oxygen environment, which the enzymes involved also produce.
A dream combination, sought for years, is a renewable power source for production, such as wind or solar, with a cheap catalyst that would drive production costs down. Last year, an MIT professor announced his progress in, “Integrating the cobalt catalyst with a solar cell to create a device that uses the sun to split water.”
Dr. Daniel Nocera, the Henry Dreyfus Professor of Energy at MIT has been working with graduate student Matthew Kanan (as of 2008) on using a new catalyst that produces oxygen gas from water; while another catalyst produces valuable hydrogen gas. According to MIT, “The new catalyst consists of cobalt metal, phosphate and an electrode, placed in water. When electricity — whether from a photovoltaic cell, a wind turbine or any other source — runs through the electrode, the cobalt and phosphate form a thin film on the electrode, and oxygen gas is produced.”
This would allow storage of the energy gained from the electricity generated by clean sources in the form of hydrogen, which could power fuel cells when the sun is down or the wind dwindles.
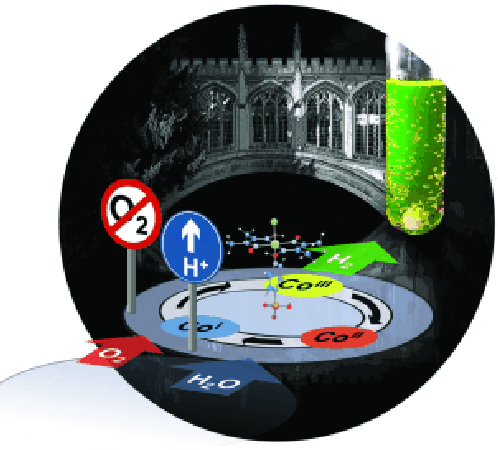
Scientists at Cambridge University in England have produced H2 from water under “real-world conditions,” according to the University. This employs an inexpensive catalyst under “industrially-relevant conditions” using pH neutral water, surrounded by atmospheric oxygen at room temperature. This might verify Dr. Nocera’s earlier efforts, and certainly shows a direction that researchers might follow.
Dr. Erwin Reisner, head of the Christian Doppler Laboratory at the University, and who incidentally did graduate work at MIT from 2005 to 2007, has come to similar conclusions as Nocera’s and says, “Our research has shown that inexpensive materials such as cobalt are suitable to [create hydrogen under those ‘industrially-relevant conditions.’ Of course, many hurdles such as the rather poor stability of the catalyst remain to be addressed, but our finding provides a first step to produce ‘green hydrogen’ under relevant conditions.”
“The results show that the catalyst works under air and the researchers are now working on a solar water splitting device, where a fuel H2 and the by-product O2 are produced simultaneously. Fezile Lakadamyali and Masaru Kato, co-authors of the study, add: ‘We are excited about our results and we are optimistic that we will successfully assemble a sunlight-driven water splitting system soon.’”
As Dr. Nocera and Dr. Reisner’s teams press on, we can hope to see an inexpensive means to generate the power we need for the future.
Funded in part by the Engineering and Physical Sciences Research Council (EPSRC), their research is available online in the journal Angewandte Chemie International Edition under the title, “Selective Reduction of Aqueous Protons to Hydrogen with a Synthetic Cobaloxime Catalyst in the Presence of Atmospheric Oxygen,” as presented in the September 10, 2012 edition.
Co-authors Fezile Lakadamyali, Dr. Masaru Kato, Dr. Nicoleta M. Muresan and Dr. Reisner share the honors.

Comments 1
The true requirement is for a way to enable distributed hydrogen production. A method that only is cost effective on a large scale builds in the cost of distributing the hydrogen. A true hydrogen system is one that can produce sufficient hydrogen to power a fuel cell car on the premises of the individual house. That has to include the means of compression or hydridization to get the hydrogen stored on board the vehicle. Simple electrolysis can meet both of these needs but it is not cheap.