Dr. Cui is at it again! In a seemingly endless stream of announcements, his work with silicon anodes keeps promising improvements in battery capacity and longevity. The Stanford professor and his team, Stanford’s National Accelerator Laboratory (Formerly the Stanford Linear Accelerator Center), and the Environmental Molecular Sciences Laboratory (EMSL) at Pacific Northwest National Laboratory all published papers on their latest joint accomplishment.
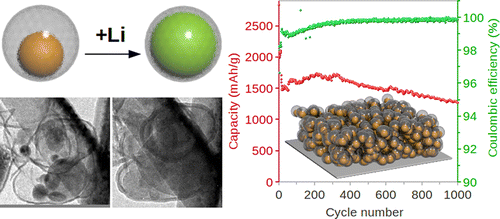
- Conceptual drawing of silicon filling carbon shell, TEM photo of actual expansion, and life cycle analysis for yolk-shell batteries
Expansion and contraction of anodes and cathodes during charging and discharging of batteries causes flexing and eventual breakdown of a battery’s internal components. Cui and other researchers have tried various strategies to mitigate or eliminate this flexing, but the latest tactic seems to promise longer battery life and greater power and energy.
Calling it a “yolk-shell structure,” researchers seal commercially available single silicon nanoparticles in “conformal, thin, self-supporting carbon shells, with rationally-designed void space between the particles and the shell.” When charged, the silicon nanoparticles swell to fill the carbon shells, just the right size to contain the expansion and not burst.
Researchers claim 2,800 milli-Amp hours per gram at 10 to 20 times normal discharge (C)*, 1,000 charge/discharge cycles so far with 74-percent capacity retention, and 99.84-percent Coulombic efficiency, or the percentage of charge retained by the battery – very high indeed for this system.
An in-situ Transmission Electron Microscope (TEM) was used to observe the changes in the silicon yolks and carbon shells during charging and discharging, showing the effects of “lithiation” or infusing the shell with charged lithium. This would normally lead to sub-nano-scale cracking of the anode, but in this case leaves the structure undamaged and ready for more full charges.
As explained in the abstract of the team’s research, “In this new design, lithium ions flow from the cathode through the electrolyte, diffuse through the carbon shells, and enter the silicon—which can hold ten times as many lithium ions as carbon alone. By leaving just the right amount of space, the lithiated silicon nanoparticles swell to fill, but not burst, the carbon shell. The result? A lithium ion battery system that compared to commercial batteries holds seven times more energy and can be discharged and recharged five times as many times before it wears out.”
“This is a very exciting development toward our goal of creating smaller, lighter and longer-lasting batteries than are available today,” Cui said.
Research findings were published by Nian L, H Wu, MT McDowell, Y Yao, C Wang, and Y Cui as “A Yolk-Shell Design for Stabilized and Scalable Li-Ion Battery Alloy Anodes” in Nano Letters and in the March 25 Nature Nanotechnology.
With anticipated results heading toward 6,000 cycles, or one full cycle each day for over 16 years, such batteries would essentially be lifetime power sources in light aircraft use. Even after all that, they would still hold 85-percent of their original design charge, good enough for use in the family EV for a few more years.
Dr. Cui is part of Amprius, a commercial enterprise dedicated to making these new cells a reality in our daily lives.
Thank you to Jeff Shingleton, co-pilot on the Green Flight Challenge Phoenix, who shared news of this work with your editor at the recent International Vintage Sailplane Meet at Harris Hill, New York.